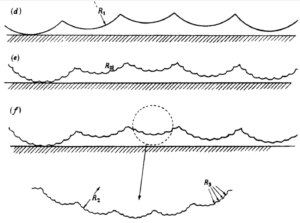
Crystallinity is a fundamental property of polymers that describes the degree of order in their molecular structure. It can have a significant impact on the tribological properties of a material in biotribological applications. The tribological properties of a material refer to its behavior during friction, wear and lubrication. In general, polymers with higher crystallinity tend to have better mechanical and thermal properties, such as increased strength and stiffness, as well as better wear resistance. This is due to the fact that the ordered arrangement of molecules in a highly crystalline polymer allows for stronger intermolecular interactions, leading to a more durable material.
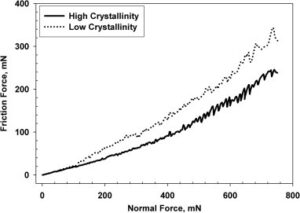
In contrast, polymers with lower crystallinity tend to have poorer mechanical properties and increased wear. The disordered arrangement of molecules in these materials leads to weaker intermolecular interactions, making them more susceptible to deformation and wear. In biotribological applications, such as artificial joint replacements, this can lead to increased wear on the implant and a shorter lifespan for the device.
Crystallinity also affects the coefficient of friction of a polymer. Polymers with higher crystallinity tend to have a lower coefficient of friction, as the orderly arrangement of molecules allows for less deformation and better sliding properties. In contrast, polymers with lower crystallinity tend to have a higher coefficient of friction, as the disordered arrangement of molecules leads to increased deformation and poorer sliding properties. This can be a significant issue in biotribological applications where low friction is desired, such as in artificial joint replacements or in biomedical devices that need to slide against living tissue.
Furthermore, Crystallinity can also have an impact on the biocompatibility of a polymer. Polymers with higher crystallinity tend to be more biocompatible, as they are less likely to cause an inflammatory response in the body. In contrast, polymers with lower crystallinity tend to be less biocompatible, as the disordered structure of the polymer can lead to an increased risk of an inflammatory response.
In conclusion, Crystallinity is a crucial property that plays a significant role in the tribological properties of a polymer, including wear resistance, coefficient of friction, and biocompatibility. In biotribological applications, such as artificial joint replacements, it is important to carefully consider the crystallinity of a polymer when selecting materials. The crystallinity of a polymer can be controlled by various methods such as thermal treatment, addition of additives, and so on. Therefore, by understanding the effects of crystallinity on the tribological properties of a polymer, scientists and engineers can make more informed decisions when designing and developing biotribological devices.
Sources:
Wang and S. Ge, “The mechanical property and tribological behavior of uhmwpe: Effect of molding pressure,” Wear, vol. 263, no. 7-12, pp. 949–956, 2007.
K. Karuppiah, A. L. Bruck, S. Sundararajan, J.Wang, Z. Lin, Z.-H. Xu, and X. Li, “Friction and wear behavior of ultra-high molecular weight polyethylene as a function of polymer crystallinity,” Acta Biomaterialia, vol. 4, no. 5, pp. 1401–1410, 2008.

This article was written by Dilesh Raj Shrestha as part of an ongoing series of scientific communications written and curated by BioTrib’s Early Stage Researchers.
Dilesh is researching the Development of 3D-printable, self-lubricated polymer composites with improved wear resistance for total joint replacement at Luleå University of Technology, Sweden.